Celiac Disease Diagnosis: Serology, Biopsy, and Gluten-Free Management
Serologic Testing: The First Step
Celiac disease affects a substantial portion of the population but remains underdiagnosed — many patients carry the diagnosis for years before it is recognized, often presenting with atypical features that do not immediately suggest malabsorption. For the gastroenterologist, primary care physician, or internist evaluating patients with unexplained iron deficiency, chronic diarrhea, or elevated transaminases, having a clear understanding of the diagnostic algorithm from serology through biopsy to molecular testing is essential for timely and accurate diagnosis.
Tissue transglutaminase IgA (tTG-IgA) is the recommended initial screening test, with sensitivity of 93-98% and specificity of 95-98%[1] in adults on a gluten-containing diet. Total serum IgA should be measured concurrently, as IgA deficiency (present in 2-3% of celiac patients versus 0.2% of the general population)[1] produces false-negative tTG-IgA results. In IgA-deficient patients, deamidated gliadin peptide IgG (DGP-IgG) or tTG-IgG should be substituted. Endomysial antibody (EMA-IgA) serves as a confirmatory test with near-100% specificity but lower sensitivity (approximately 90%) and higher cost.
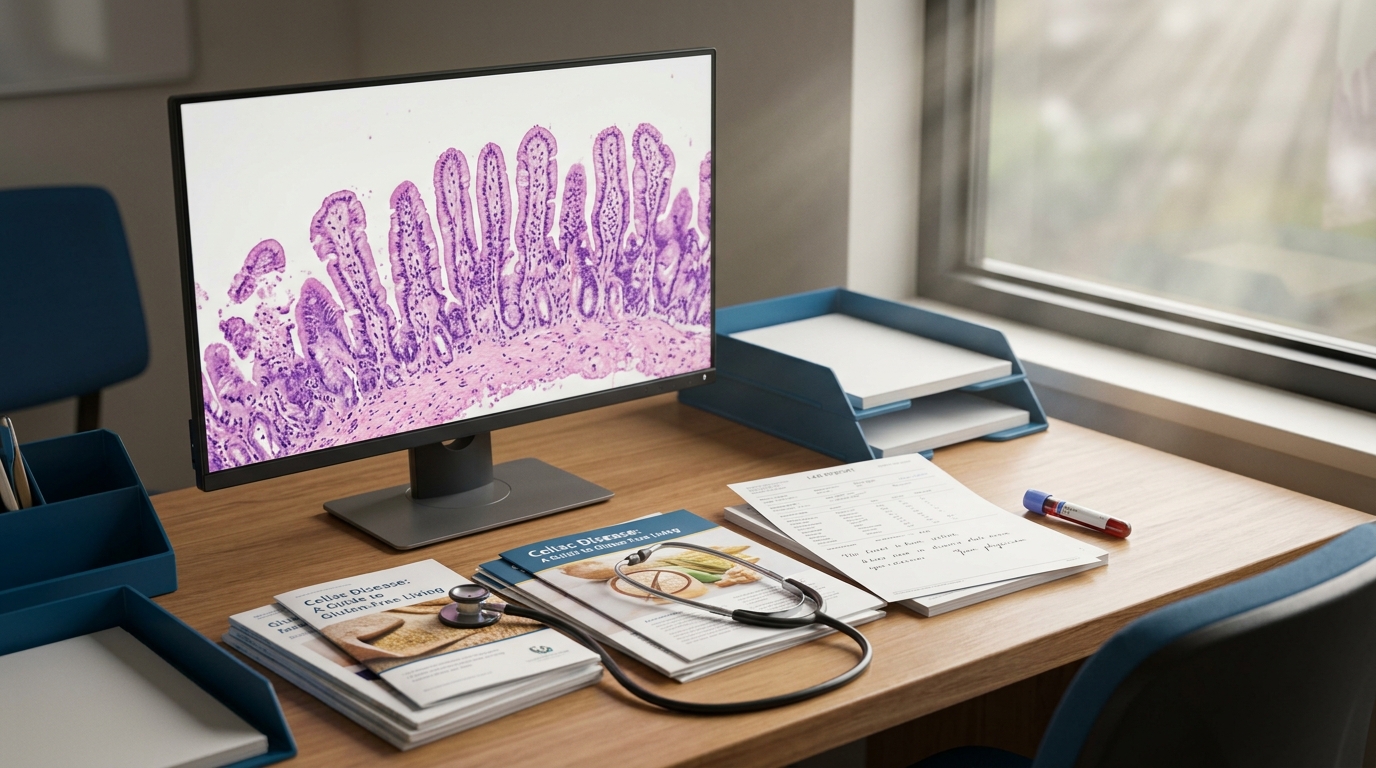
Duodenal Biopsy: Still the Gold Standard
The 2023 ACG Clinical Guideline recommends esophagogastroduodenoscopy with at least 4 biopsies from the second portion of the duodenum and 2 from the duodenal bulb[1] for all adults with positive serology. Marsh classification grades histologic severity: Marsh 1 (increased intraepithelial lymphocytes above 25/100 enterocytes), Marsh 2 (crypt hyperplasia), and Marsh 3a-c (partial to total villous atrophy). Notably, patchy mucosal involvement occurs in up to 20% of cases[1], making multiple biopsies essential to avoid sampling error.
Serology-Only Diagnosis: Pediatric Model, Adult Potential
The 2020 ESPGHAN guidelines allow biopsy-free diagnosis in children when tTG-IgA exceeds 10 times the upper limit of normal with positive EMA on a separate blood sample[2]. The ProCeDE study validated this approach in 707 children with 99.75% positive predictive value[3]. In adults, the multinational CREST trial and several retrospective analyses suggest that tTG-IgA above 10x ULN with positive EMA predicts Marsh 3 histology in over 95% of cases. However, current ACG guidelines still recommend confirmatory biopsy in adults pending further prospective data.
Gluten-Free Diet: Implementation and Monitoring
A strict lifelong gluten-free diet (GFD) remains the only established treatment, essential to prevent complications including iron deficiency anemia and bone loss. Gluten intake must be reduced below 20 parts per million to prevent mucosal damage. Dietitian consultation at diagnosis improves adherence from approximately 40% to 75% at 1 year[1]. Monitoring includes serial tTG-IgA measurements every 3-6 months, with normalization expected within 12-24 months. Persistent elevation beyond 24 months suggests ongoing gluten exposure (intentional or inadvertent) and warrants dietary reassessment before considering refractory celiac disease workup.
Emerging Therapies and Refractory Disease
Refractory celiac disease type II (RCD-II), characterized by aberrant intraepithelial lymphocyte clonality and risk of cancer surveillance implications, carries a 5-year mortality of 40-60%[1] with risk of enteropathy-associated T-cell lymphoma. Cladribine-based regimens and autologous stem cell transplantation have shown partial responses. In the pipeline, latiglutenase (glutenase enzyme) showed symptom improvement in the ALV003-1221 trial, while ZED1227 (transglutaminase 2 inhibitor) demonstrated mucosal protection in a phase 2a study (68% reduction in villous height ratio decline versus placebo, p=0.003)[4].
The Most Common Diagnostic Pitfalls
Two errors account for the majority of missed celiac diagnoses. The first is testing patients who have already started a gluten-free diet on their own. Both serology and biopsy require the patient to be consuming gluten for adequate sensitivity — a patient who has been gluten-free for weeks or months may have false-negative tTG-IgA and normal-appearing duodenal mucosa. Before testing, patients should be consuming the equivalent of at least two slices of wheat bread daily for 6-8 weeks (the "gluten challenge"). The second is failing to check total IgA alongside tTG-IgA. The clinician who orders tTG-IgA without total IgA and gets a negative result may miss celiac disease entirely in the subset of patients with selective IgA deficiency. These are simple protocol steps, but omitting them leads to diagnostic delays that can span years.
Limitations and Living with a Lifelong Diagnosis
The gluten-free diet is effective but far from simple. Cross-contamination occurs routinely in restaurants, shared kitchens, and processed foods, and even small amounts of gluten can sustain mucosal inflammation in sensitive patients. The social and psychological burden of a lifelong dietary restriction — particularly for adolescents and young adults — is often underappreciated by clinicians who focus on the nutritional and serologic aspects of management. Dietitian involvement at diagnosis is the single most impactful intervention for adherence, and periodic reassessment of dietary compliance using both serology and symptom review should be part of every follow-up visit. The pipeline therapies described above may eventually provide adjunctive protection against inadvertent gluten exposure, but a complete dietary replacement is not on the near-term horizon.
References
- ACG Clinical Guideline: Diagnosis and Management of Celiac Disease (2023) PubMed 36508693
- European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020 (Husby et al.) PubMed 31568151
- ProCeDE: Prospective Assessment of the Diagnostic Accuracy of the Biopsy-Avoidance Approach in Children PubMed 28624578
- A Randomized Trial of a Transglutaminase 2 Inhibitor for Celiac Disease (ZED1227, NEJM 2021) PubMed 34192430
Frequently Asked Questions
What is the sensitivity and specificity of tTG-IgA for celiac disease?
Can adults with celiac disease be diagnosed without biopsy?
How many duodenal biopsies are needed to diagnose celiac disease?
When should tTG-IgA normalize on a gluten-free diet?
What is the prognosis for refractory celiac disease type II?
Does dietitian consultation improve gluten-free diet adherence?
Explore This Topic in Ailva
Ailva is a free clinical intelligence platform for NPI-verified US physicians. Get evidence-based answers with verified citations from 16M+ indexed papers — plus free CME credits.

Founder of Ailva.ai | Former Director of Research and Author of 200+ Medically Reviewed Articles | Editor-in-Chief of EudaLife Magazine